Capsule Disintegration Time: A Guide to USP Standards & Testing Equipment
Dissolving a capsule is just beautiful. They have a mystifying science. A crisscrossed network established by a blend of several ingredients releases the potential content quickly when reaches the site of action. Initially, fluid enters the spongy molecules to begin dissolving phenomena. This is a simple picture of capsule dissolving. How long does it take for a capsule to dissolve? In reality, the body system is far more complicated. Before jotting down the body’s physiological response. We want to highlight this parameter from outside the body (in vitro).
| Capsule Type | USP/EP Standard | Target Dissolution Environment | AIPAK Testing Solution |
| Hard Gelatin Capsule | < 15 - 30 Minutes | Gastric Juice (Stomach) | BJ-Series Disintegration Tester |
| Vegetable (HPMC) Capsule | < 30 Minutes | Gastric Juice (Stomach) | Automatic Bath Heater System |
| Enteric Coated Capsule | > 2 Hours (Stable in Acid) | Intestinal Juice (pH 6.8) | Dual-beaker Programmable Tester |
| Softgel Capsule | < 45 Minutes | Gastric Juice (Stomach) | Heavy-duty Basket Assembly |
Table of Contents
Ⅰ.Nature Of Capsules
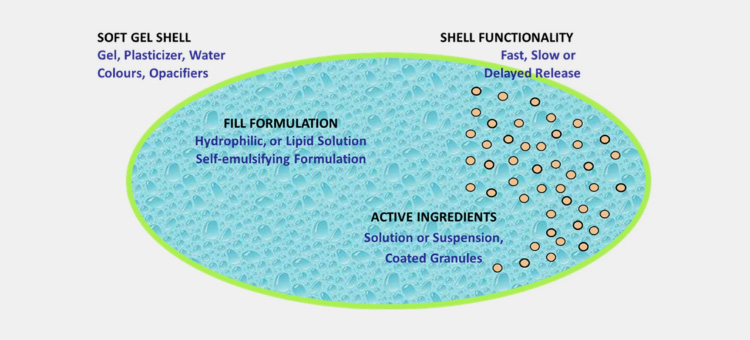
Certainly, depending upon the nature and physical characteristics of the capsule you can determine the way they dissolve. As you know that capsules are of two basic types such as soft gelatin capsules and hard gelatin capsules. Soft gelatin capsules have a thicker shell and exhibit high elasticity due to the high concentration of plasticizers so it takes a longer time to rupture than hard gelatin capsules.
On another side, hard gelatin capsules are thinner and more rigid so the ratio of dissolution is comparatively more rapid than soft gelatin.
Ⅱ.Characteristic Of Dissolving Material
Before dissolving the capsule, you must determine the quality of the chemical and physical properties of the fill material as well. This is an important parameter because hydrophobic solutions (oil-loving) such as neat oil, miscible oils, or APIs in oils can’t favor dissolving in water.
Similarly hydrophilic (water-loving) content is a combination of water-miscible liquids that can be dissolved in the water-miscible vehicle.
Ⅲ.How To Dissolve Capsules
Capsules are made up of gelatin or plant-based materials. These capsules rapidly disintegrate by absorbing water. Dissolution of capsules is optimized at 37°C which is normal body temperature. Capsules are the preferred form of medication as these quickly disintegrate in the body providing quick relief.
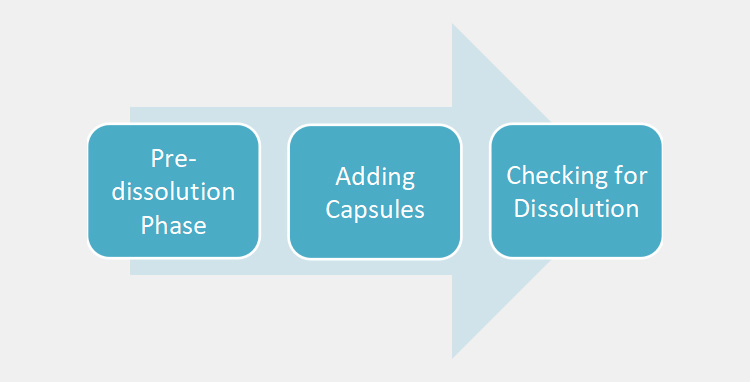
Simple steps for dissolving capsules in-vitro are detailed below for your ease:
Pre-dissolution Phase
- Gather all the supplies like cups, napkins, etc for dissolving capsules. Place the absorbent material or paper towels on the working area for absorbing spills
- Put on gloves after washing your hands. Set the capsule container in the working area. Add about some ml of water/solvent to the container.
Adding Capsules
- With the help of tweezers, pick up capsules. Place the capsule in a measuring cup containing water.
- Lightly swirl cup forth and back
Checking for Dissolution
- Leave it for some minutes. At this time, you will see partial dissolution.
- It will take 20-30 minutes for capsules to dissolve completely. (Note: the dissolution time may vary from type and nature of capsule to capsule).
Ⅳ.How Long Does It Take For A Capsule To Dissolve?
Gelatin capsules both hard gel and softgel rapidly dissolve in water in a few minutes. Standard gelatin capsules are usually completely dissolved in water within 20-30 minutes for their submergence.
Vegetarian capsules containing modified cellulose take quite a time about 30 to 60 minutes for complete dissolution.
STEPS INVOLVED IN DISSOLVING CAPSULE
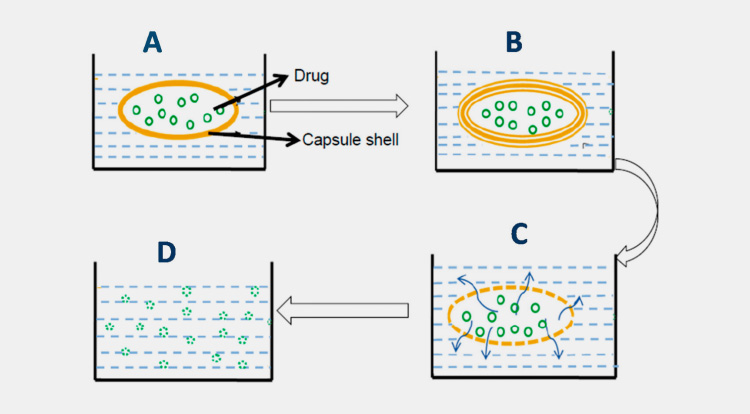
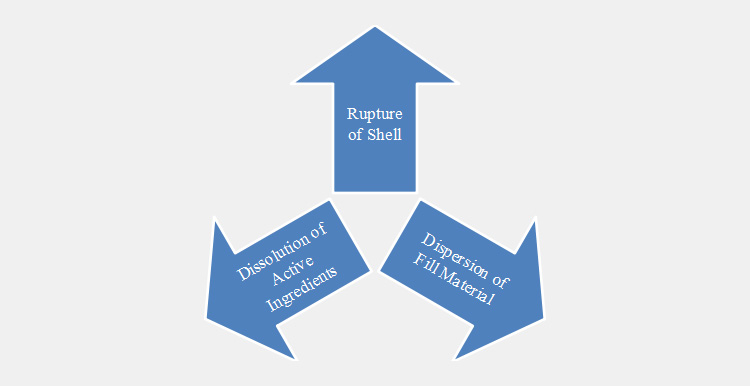
The gelatin capsule passes through the following basic phases of dissolutions.
Swelling
When dissolving a capsule in a dissolution medium first alternation in its structure begins with the swelling of gelatin molecules
Rupture
Swelling results in the rupturing of gelatin shell followed by releasing and dispersing of filling material.
Dissolution of Ingredients
In this phase, active ingredient(s) are dissolved in the dissolution medium. Such processes occur in a gradual series; however, the dissolution rate varies from material to material.
Ⅴ.Industrial Equipment Used To Dissolve A Capsule
Discussed below are the following equipment approved by USP (United States PHARMACOPEA) utilized in the industrial sector for determining the dissolving characteristics of capsules.
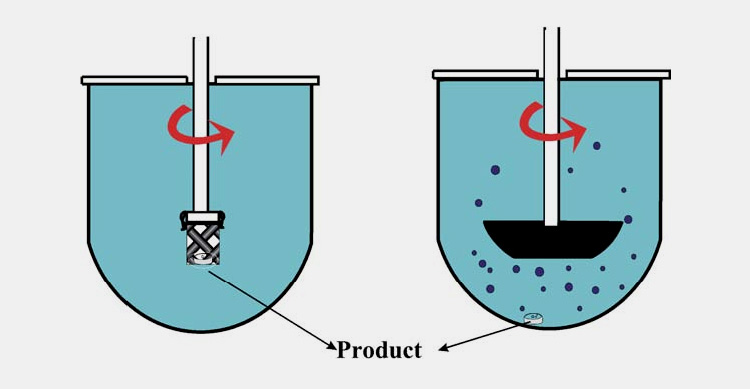
USP APPARATUS 1 – ROTATING BASKET
The apparatus is used to dissolve the capsule to prevent them from free-floating in the media. Basket apparatus may not be suitable for certain capsules as ruptured material clogs in the mesh and for hydrophobic filled ingredients; the oil phase released from the capsule may not properly be dispersed into the medium via mesh. A large-sized mesh is required to overcome the problem.
USP APPARATUS 2- ROTATING PADDLES
The apparatus does not have mesh to clog to the material but it cannot prevent the capsule from floating. Sinkers are used to prevent the problem and ensure a better contact with the dissolution medium, keeping the capsule remain at the bottom of the container.
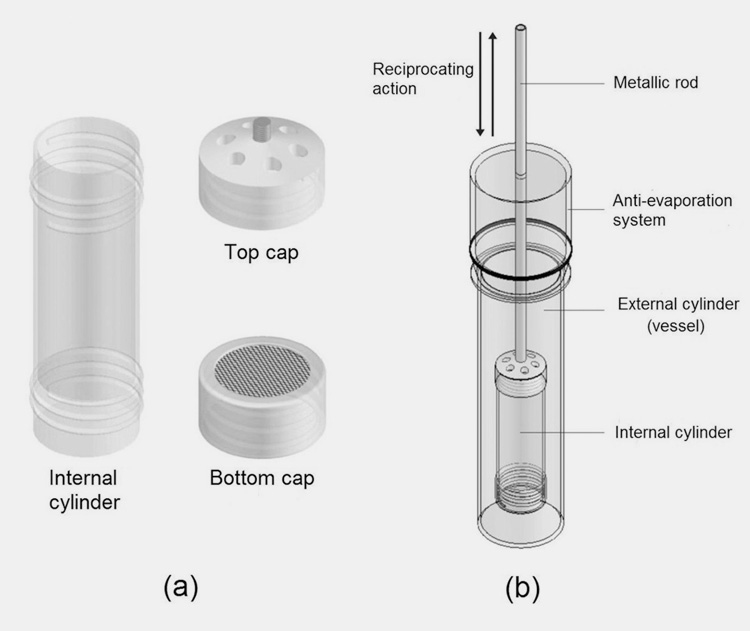
USP APPARATUS 3- RECIPROCATING CYLINDER
Like basket apparatus, it encloses the capsule, but a change in the mechanism ensures a different hydrodynamic than apparatus 1 and 2. This apparatus is useful to determine the dissolution of targeted or modified release drugs. The apparatus offers an intense turbulent environment that is useful in the dispersion of hydrophobic filler material.
USP APPARATUS 4- FLOW-THROUGH CELL
The apparatus encloses the capsule accompanied by a filter. This apparatus is designed for lipid-filled gelatin capsules and can be set up for the high or low volume of media suitable for the poorly soluble active ingredient(s).
Ⅵ.Different Factors That Affect A Capsule To Dissolve
There are several factors that affect the dissolution of capsules. These are:
Temperature
Dissolution of HPMC capsules is temperature independent. These capsules have the same rate of dissolution at temperatures between 10 -55°C.
But the dissolution of gelatin and PEG capsules is temperature-dependent. These capsules dissolve slowly at 30°C.
Size
Larger the size longer the dissolution time. A capsule of size 0 disintegrates more rapidly than of size 3.
pH
The pH of the solution also affects the dissolution of capsules. A solution having high ionic strength decreases the rate of dissolution.
Shell Material
It is seen that there is a considerable reduction in dissolution time if shell of capsules have more moisture content as moistened shell aids in polymer hydration.
Cross-Linking
Cross-linking between encapsulated material and capsule coating results in the formation of water-insoluble protein on the outer or inner surface of the capsule shell. This causes difficulty in the disintegration of capsules.
AIPAK Expert Insight: Why Consistent Fill Weight is Critical for Dissolution
"In our laboratory trials, we've found that over-compressing powder during the filling process can significantly delay capsule disintegration. If your powder plug is too dense, the solvent cannot penetrate the capsule shell effectively. At AIPAK, our Automatic Capsule Filling Machines utilize tamping pin technology with ±1% weight precision, ensuring that every capsule has the ideal porosity for consistent, reliable dissolution according to your specific formulation requirements."
Ⅶ.Precautions While Dissolving Capsules
Following precautions must be taken while dissolving capsules as safety comes first.
- Wear gloves and a mask to avoid contact with hazardous chemicals.
- Clean the workbench before and after dissolving capsules to prevent the risk of contamination.
- Wear overall for averting accidental spillage.
- Dissolve capsules in the uncluttered work area that has proper ventilation
- Rinse the measuring cup with soapy water after dissolution and allow it to air dry.
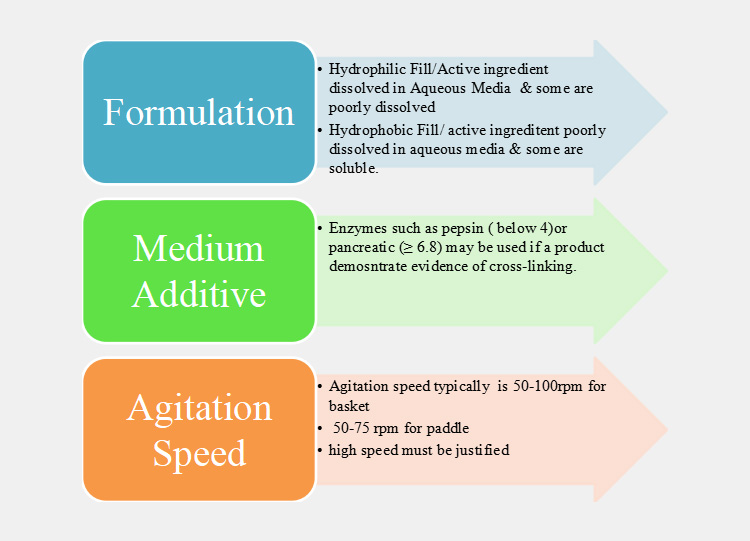
Ⅷ.Suggestions For The Dissolving The Capsule
Based on the consideration above, the chart illustrates the possible starting points that must be considered before dissolving a capsule:
Troubleshooting Common Capsule Disintegration Failures
In a pharmaceutical environment, a "Disintegration Failure" can lead to entire batch rejections. At AIPAK, we help our clients identify the root causes of these delays through engineering and process optimization:
1. Excessive Use of Lubricants (The Hydrophobic Barrier)
-
The Problem: Over-using Magnesium Stearate or other lubricants can coat the powder particles with a hydrophobic (water-repelling) layer. This barrier prevents gastric fluids from penetrating the capsule content, significantly slowing down dissolution.
-
AIPAK Solution: We recommend optimizing your blending cycle with our Bin Mixers to ensure uniform lubricant distribution without over-blending, which preserves the ideal disintegration profile.
2. Improper Storage & Environmental Humidity (Cross-linking Risk)
-
The Problem: Gelatin capsules are highly sensitive to humidity. High moisture levels can trigger "cross-linking," an irreversible chemical reaction that creates a rubbery, insoluble film on the capsule shell. Conversely, low humidity makes shells brittle and prone to cracking.
-
AIPAK Solution: This is why stable environment control is non-negotiable. As part of our Cleanroom Engineering services, we design HVAC systems that maintain precise RH (Relative Humidity) levels, ensuring your capsules remain stable from filling to final packaging.
3. Inconsistent Shell Quality (The Foundation of Success)
-
The Problem: Lower-grade empty capsules often have inconsistent wall thickness or impurities in the raw gelatin/HPMC, leading to erratic disintegration times within the same batch.
-
AIPAK Solution: We always advise using high-quality Empty Capsules that meet international pharmacopeia standards. Combined with AIPAK’s Automatic Capsule Filling Machines, which ensure precise tamping and closing, you can achieve 100% batch-to-batch consistency in dissolution tests.
Conclusion
"Struggling with Inconsistent Capsule Dissolution Rates?" Don't let batch failures ruin your production timeline. Whether you need high-precision Capsule Filling Machines or USP-compliant Disintegration Testers, AIPAK offers the turnkey laboratory and production solutions you need. Contact our technical team today for a custom equipment proposal or a 3D layout of your QC laboratory. Request a Quote for Capsule Testing & Production Equipment.
Don't forget to share this post!
Capsule Filling Machine Related Posts
Capsule Filling Machine Related Products
Capsule Filling Machine Related Videos
CONTACT US
Tell us your raw material and project budget to get quotations within 24 hours.
WhatsApp Us: +86 156 0710 8630

Want the best price & newest pharmaceutical machinery buying guide,tips and trends sent straightly to your box?Sign up for Aipak’s monthly newsletter,we’re free for your consultation and Offer you the most suitable solutions!
The Buyer's Guide
- Capsule Filling Buyer's Guide
- Blister Packaging Buyer's Guide
- Tablet Counting Buyer's Guide
- Tube Filling Buyer's Guide
- Cartoning Buyer's Guide
- Gummy Making Buyer's Guide
- CO2 Extraction Buyer's Guide
- Empty Capsules Buyer's Guide
- Suppository Filling Buyer's Guide
- Tablet Coating Buyer's Guide
- Tablet Press Buyer's Guide
- Softgel Encapsulation Buyer's Guide
Most Popular
- 7 Importance Of Pharmaceutical Packaging In Different Applications You Must Know
- 6 Advantages You Must Know About Tablet Counting Machine
- 8 Advantages of Blister Packaging You Must Know
- 6 Critical Applications of Automatic Capsule Filling Machine
- 6 Stations You must Know to Improve the Filling Quality of Automatic Capsule Filling Machine